Difference Between a Vented Ball Valve and a Standard Ball Valve
Posted by Russ Bailey on Jul 11th 2016

While the difference between a vented ball valve and a standard ball valve may seem obvious on the surface of what constitutes their individual functions, that distinction becomes far more critical when we introduce the factor of cryogenic processing. In such an application, it is important to know more than just the surface level distinctions, and it is crucial to understand--more importantly--why they are different. So, let's examine why a vented ball valve is used for cryogenics and what advantages it possess when supporting cryogenic applications.
Vented Ball Valve vs. Standard Ball Valve
A vented ball valve (often referred to as a cryogenic valve) differs significantly from a standard ball valve, because it has a small hole drilled into its upstream side. A standard ball valve, which is comprised of a housing, a ball featuring a bore through which material may flow, a stem controlling the ball position, and seals where the ball rests between, does not have a visible hole in the ball when the valve is in the closed position. Otherwise (from at least a surface level), the two valves are identical, and operate in the same way. Turned one way, the bore of the ball allows upstream flow--the permission of travel from one end of the valve (inlet), to the outlet. Turned the other way, travel of material is completely halted.
A standard ball valve--in its simple yet ingenious design--is perfect for countless applications involving various materials. When closed, the ball creates a snug seal. A standard ball valve is optimal for water and similar fluid applications. But, as in the case of cryogenic processing, it can cause leaking, valve failure, or even explosion, due to internal pressure arising from the natural process in which the cryogenic process heats up, turning into a gas, and expands inside of the valve itself. A vented ball valve eliminates this unwanted pressure inside of the valve by venting the pressure through the "vent" on the ball.
Now that we understand how this pressure develops, and how a vented ball valve counteracts this problem, we can now understand the answer to the question of, "Why is a vented ball valve needed?" When transporting a cryogenic substance like Liquefied Natural Gas--a liquid that wants to be a gas--extremely cold temperatures must be applied, as heat spikes can result in disaster. In other words, heat causes pressure. These temperatures fall well below refrigeration, which begins at -45 degrees Fahrenheit. This extreme cold presents another problem, as it causes the shrinkage and cracking of seals. To compensate, special seals, designed to handle temperatures -100 degrees F or colder, must be employed.
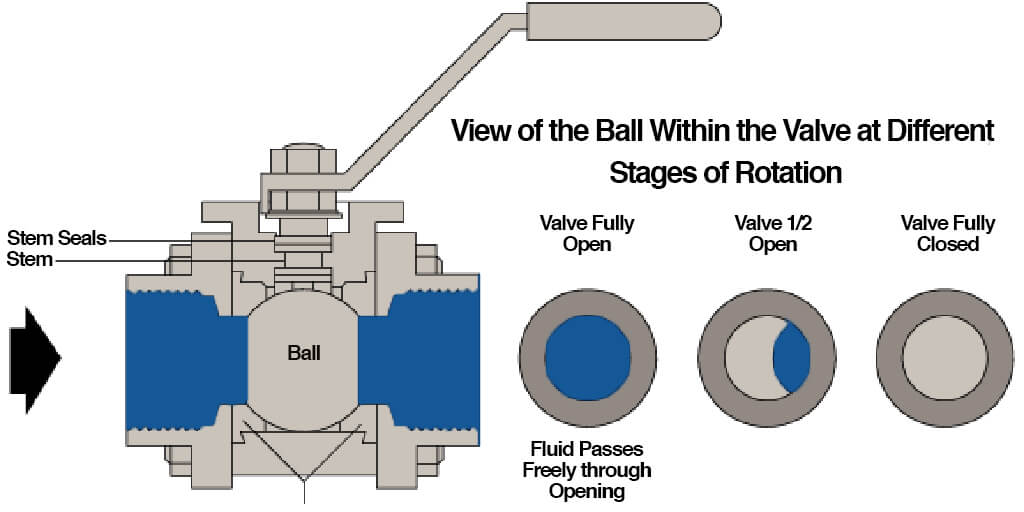
Maintaining Temperature and Pressure
Temperature and pressure fluctuations in a line tend to occur when media enters a valve. This is especially true of cryogenic fluids, as maintaining their liquid state depends on maintaining their temperature, which is harder to control when movement is halted. Without sounding too technical, it is greatly effected by aspects of kinetic energy and thermal expansion, and in the case of cryogenic material, heat causes pressure, and this pressure must be relieved in order to prevent leakage or valve failure. This is why, the ball must be vented in cryogenic applications, because a non-vented or standard ball valve will simply fail under such intense applications.
Where to Shop Valves
Whether you need to purchase a specialized valve like a cryogenic valve or a simple standard ball valve, you should always consider buying from a knowledgeable distributor. The pros at ValveMan.com have been engineering specialty valves for over fifty years and their online valve store has a wide selection of vented ball valves that are ready to order. We value our customers, pride ourselves on our customer service, and want to provide valuable educational resources that help you make the right choice when selecting a valve. We do this because we are passionate about valves. In fact, unlike our competition, valves are all we sell, and we make valve shopping as easy as possible for you.



